You’ve stood in the shampoo aisle reading bottle backs until your eyes blur. The ingredient lists stretch on forever, filled with chemical names you can’t pronounce and claims you’re not sure you trust. You want to make informed choices, but how are you supposed to know if ‘sodium laureth sulfate’ is the villain or if ‘hydrolyzed wheat protein’ actually does anything?
Here’s what makes this harder in the Gulf region: the water itself changes what you need from a shampoo. Ingredients that work beautifully in soft water can fail completely when you’re washing with water that’s loaded with calcium and magnesium. That sulfate-free shampoo everyone raves about? It might not cleanse effectively enough to remove the mineral film that builds up here.
This article contains affiliate links. See our affiliate disclosure for details.
Learning to read ingredient labels isn’t about memorizing chemistry. It’s about understanding what each category of ingredients does and whether it addresses your specific environmental challenges. Once you know what to look for, you can spot the difference between marketing language and formulations that actually work in hard water conditions.
Key Takeaways
• Ingredient lists are ordered by concentration: the first five ingredients make up the majority of the formula
• Sulfates aren’t universally bad, but the type matters: sodium lauryl sulfate is harsher than sodium laureth sulfate or gentler alternatives
• Silicones create a protective coating but require stronger cleansing to remove, especially in hard water where mineral buildup compounds the issue
• Chelating agents like EDTA, citric acid, and phytic acid are critical in hard water areas because they bind to minerals and prevent buildup
• Water-soluble ingredients appear near the top of the list, while oils and heavy conditioners appear lower, indicating their concentration and function
How Ingredient Lists Are Actually Organized
Ingredient lists follow a legal requirement: they’re listed in descending order by weight. The first ingredient is present in the highest concentration, the second ingredient is next, and so on. This continues until you reach ingredients present at less than 1% concentration, which can then be listed in any order.
For most shampoos, water (aqua) is the first ingredient because it makes up 60-80% of the formula. The next four to six ingredients are where the real action happens. These are your cleansing agents, conditioning ingredients, and functional additives. Everything after that is present in relatively small amounts, which doesn’t mean it’s unimportant, but it does mean it’s not the primary character in the formula.
This ordering system tells you something critical: if a brand advertises ‘infused with argan oil’ but argan oil appears as the 23rd ingredient, you’re getting a trace amount. The marketing might emphasize it, but the formula doesn’t rely on it. Conversely, if you see ‘disodium EDTA’ as the fifth or sixth ingredient, that’s a chelating shampoo designed to address hard water, not just a formula with a token amount added.
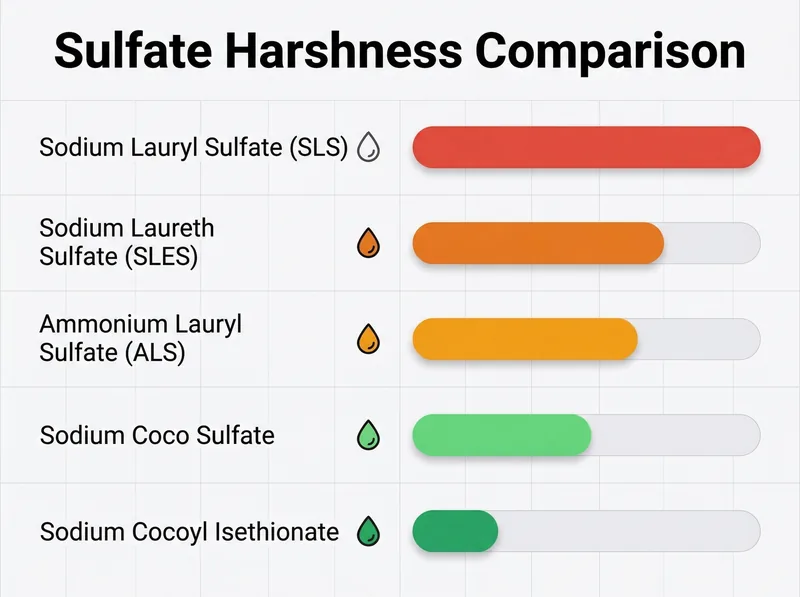 Common sulfate types ranked by harshness, from sodium lauryl sulfate (strongest) to gentler alternatives like sodium cocoyl isethionate.
Common sulfate types ranked by harshness, from sodium lauryl sulfate (strongest) to gentler alternatives like sodium cocoyl isethionate.
Sulfates: Understanding the Different Types
Sulfates are surfactants, which means they reduce the surface tension between water and oils so you can rinse away sebum, product buildup, and environmental residue. They’re not inherently harmful, but they vary significantly in how aggressively they cleanse and how much they can strip natural oils.
Sodium lauryl sulfate (SLS) is the strongest and most drying. It’s effective, which is why it appears in many clarifying shampoos, but it can be too harsh for daily use, especially if you have color-treated, curly, or chemically processed hair. Sodium laureth sulfate (SLES) is slightly gentler because it’s been ethoxylated, a process that makes the molecule larger and less penetrating.
Gentler alternatives include sodium cocoyl isethionate, sodium lauroyl methyl isethionate, and cocamidopropyl betaine (technically an amphoteric surfactant, not a sulfate, but often grouped with ‘gentle cleansers’). These are derived from coconut oil and cleanse effectively without the aggressive stripping action. In hard water, you actually need some cleansing strength because you’re fighting mineral deposits, not just oil. A too-gentle shampoo will leave you with buildup.
The research on surfactants shows that the concentration matters as much as the type. A shampoo with 15% SLS is harsher than one with 8% SLES, but a formula with 2% SLS as a secondary surfactant might be milder than one with 12% of a ‘gentle’ alternative.
Silicones: When They Help and When They Hinder
Silicones are synthetic polymers that coat the hair shaft, creating a smooth, shiny surface and reducing friction. They’re in everything from shampoos to heat protectants, and they’re not the enemy they’re often made out to be. The issue is that some silicones are water-soluble and rinse out easily, while others are not and require stronger cleansing to remove.
Dimethicone, cyclomethicone, and amodimethicone are common non-water-soluble silicones. They build up over time, especially in hard water where mineral deposits add another layer of coating. If you use products with these silicones, you need a shampoo with enough cleansing power to remove them, or you’ll end up with hair that feels waxy and looks dull.
Water-soluble silicones include dimethicone copolyol and PEG-modified silicones (anything with ‘PEG’ in the name). These rinse out with water alone, so they won’t cause buildup even if you use a gentler shampoo. If you’re in a hard water area and want the benefits of silicones without the buildup risk, look for these instead.
There’s a middle category called ‘evaporating silicones’ like cyclotetrasiloxane and cyclopentasiloxane. These provide temporary smoothing and then evaporate, leaving no residue. They’re useful in styling products but less common in shampoos. The key is knowing which type you’re using so you can match your cleansing routine accordingly.
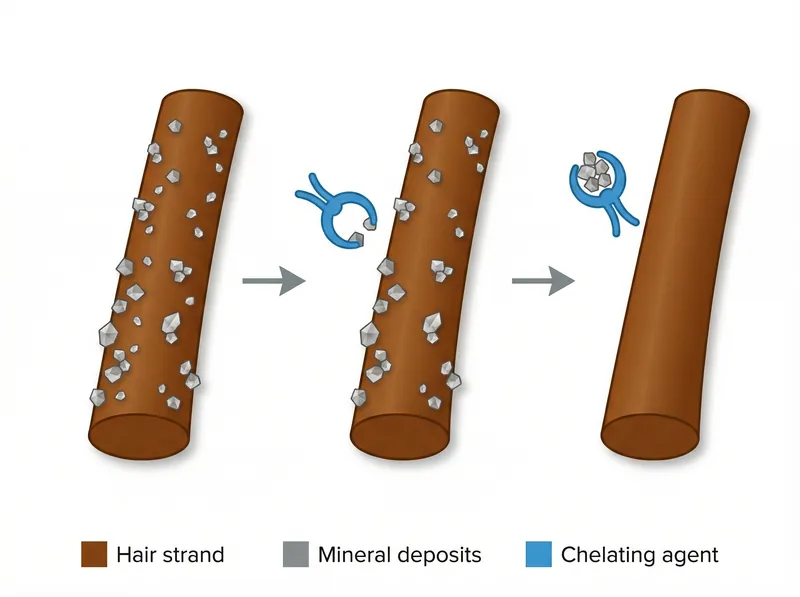 Chelating agents like EDTA and citric acid bind to mineral deposits, allowing them to be rinsed away instead of accumulating on hair.
Chelating agents like EDTA and citric acid bind to mineral deposits, allowing them to be rinsed away instead of accumulating on hair.
Chelating Agents: The Hard Water Essential
If you live in the Gulf region, chelating agents should be non-negotiable in your shampoo. These ingredients bind to calcium and magnesium ions in hard water, preventing them from depositing on your hair and allowing them to be rinsed away. Without chelating agents, minerals accumulate in a film that makes hair feel rough, look dull, and resist styling.
The most common chelating agents are disodium EDTA, tetrasodium EDTA, citric acid, and phytic acid. EDTA (ethylenediaminetetraacetic acid) is the strongest and most effective. Studies on chelation chemistry show it can bind multiple mineral ions simultaneously, making it particularly useful in very hard water.
Citric acid is gentler and often appears in ‘natural’ formulations. It works well in moderately hard water but may not be strong enough if your water has extremely high mineral content. Phytic acid, derived from rice bran or corn, is another natural option that’s gaining popularity. It’s effective but less common than EDTA or citric acid.
A chelating shampoo like Regrowth+ is formulated specifically for hard water conditions, combining chelating agents with balanced cleansing to remove mineral buildup without over-stripping. You’ll see the chelating ingredient listed in the first 10 ingredients if it’s present in a functional concentration, not just a token amount.
Conditioning and Moisturizing Ingredients
Shampoos aren’t just about cleansing. Most contain conditioning agents to counteract the drying effects of surfactants. These appear lower in the ingredient list but play an important role in how your hair feels after washing.
Cationic surfactants like behentrimonium chloride and cetrimonium chloride are conditioning agents that carry a positive charge, which helps them adhere to the negatively charged hair shaft. They’re more common in conditioners but appear in some shampoos, especially 2-in-1 formulas. In hard water, these can sometimes interact with minerals and cause buildup, so they’re less ideal than other conditioning options.
Humectants like glycerin, panthenol (provitamin B5), and hyaluronic acid attract water to the hair shaft, providing hydration without weight. These are beneficial in dry climates where humidity is low and hair tends to lose moisture. Glycerin appears in many shampoos because it’s inexpensive and effective, but it can feel sticky in very humid conditions.
Proteins like hydrolyzed wheat protein, silk amino acids, and keratin help repair damaged hair by temporarily filling in gaps in the cuticle. They’re useful if you have chemically treated or heat-damaged hair, but they can build up if overused, especially in hard water. Look for them in the middle to lower portion of the ingredient list for a functional amount without overload.
Preservatives and pH Adjusters: The Necessary Background Players
Every water-based product needs preservatives to prevent bacterial and fungal growth. The most common are phenoxyethanol, methylisothiazolinone, and parabens (methylparaben, propylparaben). These typically appear near the end of the ingredient list because they’re effective at low concentrations (usually 0.5-1%).
Parabens have been controversial, but the FDA has found no evidence they’re harmful at the concentrations used in cosmetics. Phenoxyethanol is a common alternative in ‘paraben-free’ formulas. Methylisothiazolinone is effective but can be sensitizing for some people, especially in leave-on products (though it’s less of an issue in rinse-off shampoos).
pH adjusters like citric acid, lactic acid, and sodium hydroxide ensure the shampoo stays at an optimal pH (usually 4.5-6.5 for hair products). Hair’s natural pH is slightly acidic, around 4.5-5.5, and maintaining this helps keep the cuticle smooth and closed. If a shampoo is too alkaline, it can cause the cuticle to swell and roughen, leading to frizz and damage.
You’ll also see thickeners like xanthan gum, carbomer, or sodium chloride (table salt) near the end of the list. These control the texture and viscosity of the shampoo but don’t affect hair health. They’re formulation necessities, not active ingredients.
Fragrances and Extracts: Separating Function from Marketing
Fragrance (or ‘parfum’) is a catch-all term that can represent dozens of individual chemicals. Brands aren’t required to disclose what’s in their fragrance blend because it’s considered proprietary. If you have sensitive skin or scalp, fragrance is a common irritant, and fragrance-free formulas might be worth seeking out.
Plant extracts like chamomile, aloe vera, green tea, and rosemary often appear near the end of ingredient lists. They can provide antioxidant benefits or soothing properties, but in the small concentrations typically used, their impact is minimal. They’re not harmful, but they’re also not the reason a shampoo works or doesn’t work. Marketing often emphasizes these ‘natural’ ingredients while the actual functional ingredients go unmentioned.
Essential oils are another category that sounds appealing but can be problematic. Tea tree oil, peppermint oil, and lavender oil have antimicrobial properties and can provide a pleasant scent, but they can also be irritating, especially if you have a sensitive scalp or conditions like seborrheic dermatitis. They’re not inherently bad, but they’re not universally beneficial either.
The clean beauty movement has created a perception that ‘natural’ ingredients are always better, but this isn’t supported by evidence. Synthetic ingredients are often more stable, effective, and less likely to cause allergic reactions than plant-derived alternatives. What matters is whether the ingredient is safe and functional, not whether it came from a plant or a lab.
Red Flags: Ingredients That Signal Low-Quality Formulations
While most ingredients are safe and functional, some signal that a formula is poorly designed or uses outdated chemistry. Sodium chloride (salt) as a primary thickener rather than a secondary one can be drying and irritating. If it appears in the first 10 ingredients, the formula is likely relying on salt to create viscosity instead of using more sophisticated thickening agents.
Formaldehyde-releasing preservatives like DMDM hydantoin, diazolidinyl urea, and quaternium-15 are effective but can cause sensitivity in some people. They’re less common now than they were a decade ago, but they still appear in budget shampoos. If you have scalp irritation or contact dermatitis, these are worth avoiding.
Alcohol (listed as ‘alcohol denat.’ or ‘SD alcohol’) in high concentrations can be extremely drying. A small amount is fine and often used as a solvent for other ingredients, but if it appears in the first half of the ingredient list, the formula may be too harsh, especially for dry or damaged hair.
Finally, be cautious of shampoos with an excessive number of ingredients (more than 40-50). This often indicates a formula that’s trying to do too many things at once or one that’s been heavily modified with marketing-driven additions. A well-designed shampoo can be effective with 20-30 ingredients. More isn’t better if the additions don’t serve a clear functional purpose.
Building Your Ingredient Literacy Over Time
You don’t need to memorize every ingredient or understand complex chemistry to make better choices. Start by identifying the first 10 ingredients in any shampoo you’re considering. These make up the bulk of the formula and determine how it will perform.
Ask yourself: What’s the primary surfactant? Is there a chelating agent? Are there heavy silicones or conditioning agents that might build up? Does the formula match your water conditions and hair needs? These questions will guide you toward products that work in your environment, not just products with appealing marketing.
Over time, you’ll start recognizing patterns. You’ll notice that shampoos you love tend to share certain ingredients, while ones that don’t work for you have others in common. This personal ingredient database is more valuable than any generic ‘good’ or ‘bad’ ingredient list because it’s based on your specific hair and environment.
Understanding how mineral buildup affects hair will also help you interpret ingredient lists. If you’re dealing with hard water, you now know to prioritize chelating agents and sufficient cleansing power over ‘gentle’ formulas that might not remove mineral deposits. That’s the kind of informed decision-making that ingredient literacy enables.
References
- Safety Assessment of Alkyl Sulfates as Used in Cosmetics - PubMed Central
- Chelating Agents in Cosmetic Formulations: Mechanisms and Applications - ScienceDirect
- Parabens in Cosmetics: Safety and Regulatory Status - U.S. Food and Drug Administration
- Silicones in Personal Care Products: Types and Functions - Personal Care Products Council
