You’ve been following the Curly Girl Method religiously. You avoid sulfates, you deep condition weekly, you never touch a heat tool. But your curls still feel coated, crunchy, and somehow both greasy and dry at the same time. Here’s what the curl community doesn’t always mention: some CGM-approved ingredients become actively harmful when you’re washing your hair in hard water.
The mineral content in Gulf water, desalinated supplies, and other hard water regions changes how ingredients interact with your hair. Products that work beautifully in soft water areas can create stubborn buildup, block moisture, and damage curl structure when calcium and magnesium are in the mix. This isn’t about product quality. It’s about chemistry. This article contains affiliate links. See our affiliate disclosure for details.
Understanding which ingredients amplify hard water damage gives you the power to make informed choices. Some of these ingredients appear in products marketed specifically to curly hair. Others hide under names that sound harmless. Let’s break down what actually happens when these ingredients meet mineral-heavy water, and what to look for instead.
Why Hard Water Makes Certain Ingredients Worse
Hard water contains dissolved minerals, primarily calcium and magnesium ions. When these minerals encounter certain ingredients in your hair products, they form insoluble compounds that attach to your hair shaft. The result is a coating that won’t rinse away with water alone.
This matters more for curly hair because of structure. Each twist and turn in a curl creates more surface area for mineral deposits to cling to. The raised cuticle pattern that gives curls their texture also provides more attachment points for buildup. What might cause minor dullness on straight hair can completely destroy curl definition.
Research published in the Journal of Cosmetic Science found that hard water significantly increases the negative effects of certain surfactants and conditioning agents. The minerals don’t just sit on top of these ingredients. They bond with them, creating new compounds that are harder to remove than either substance alone.
The environmental factor matters too. In regions with consistently hard water, this buildup is cumulative. Each wash adds another layer. Within weeks, you’re dealing with a mineral-product hybrid coating that no amount of conditioning can penetrate. Your curls can’t absorb moisture because there’s a barrier in the way.
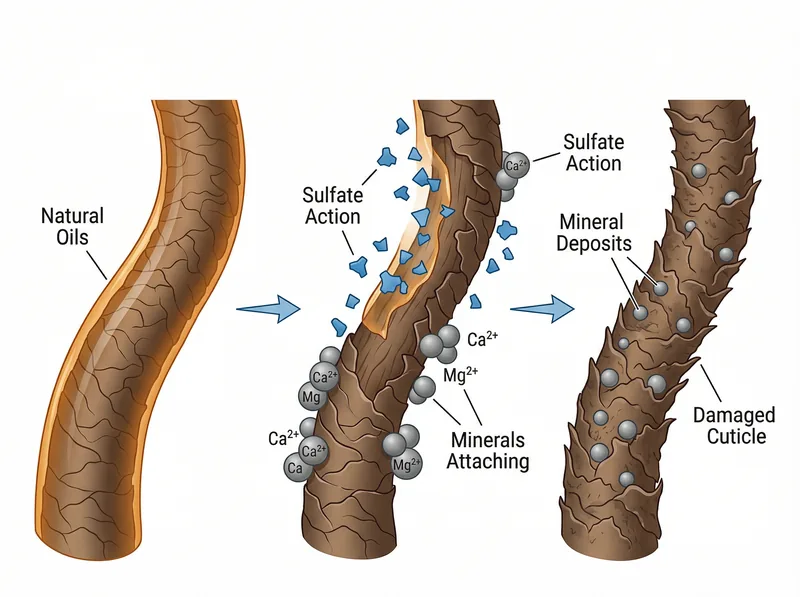 Sulfates strip natural oils while leaving mineral deposits behind, creating a cycle of dryness and buildup that’s especially damaging to curl structure.
Sulfates strip natural oils while leaving mineral deposits behind, creating a cycle of dryness and buildup that’s especially damaging to curl structure.
Sulfates: The Harsh Strip That Leaves Minerals Behind
Sodium Lauryl Sulfate (SLS) and Sodium Laureth Sulfate (SLES) are powerful cleansing agents that strip everything from your hair, including the natural oils that protect curl structure. In soft water, this is already problematic. In hard water, it becomes destructive.
Here’s the mechanism: sulfates remove your hair’s protective sebum layer, leaving the cuticle exposed and vulnerable. When hard water minerals encounter this stripped surface, they attach directly to the hair shaft. The sulfates rinse away. The minerals don’t. You’re left with dry, mineral-coated hair that feels like straw.
A study in Colloids and Surfaces demonstrated that sulfate-based cleansers in hard water conditions create significantly more residue than milder surfactants. The calcium and magnesium ions precipitate out of solution and bond with the damaged cuticle. This is why your hair feels worse after washing, not better.
What makes this particularly insidious for curly hair: the damage is progressive. Each sulfate wash strips more protective oil and deposits more minerals. Your curls become increasingly porous and prone to breakage. Many women in the Gulf region report that their hair texture changed within months of moving here. This sulfate-mineral interaction is often the culprit, not the heat or humidity alone.
![]() Non-water-soluble silicones create an impermeable barrier that traps minerals underneath, leading to progressive damage that moisturizers can’t penetrate.
Non-water-soluble silicones create an impermeable barrier that traps minerals underneath, leading to progressive damage that moisturizers can’t penetrate.
Non-Water-Soluble Silicones: The Trap That Seals Damage In
Dimethicone, Amodimethicone, Cyclomethicone. These silicones create a glossy coating that initially makes hair look shiny and feel smooth. The curl community has debated silicones for years. But in hard water conditions, there’s no debate. They’re a disaster.
Non-water-soluble silicones form an impermeable barrier on your hair shaft. When you’re washing in hard water, minerals deposit on your hair before the silicone layer forms. Then the silicone seals those minerals underneath. You’ve created a permanent coating that traps damage inside and blocks moisture from getting in.
This explains why your deep conditioning treatments stopped working. The silicone layer prevents the conditioning agents from penetrating your hair shaft. You’re essentially conditioning the silicone coating, not your actual hair. Meanwhile, the minerals trapped underneath continue to change your hair’s protein structure and damage the cuticle from within.
The only way to remove non-water-soluble silicones is with sulfates or a chelating agent. If you use sulfates, you’re back to the mineral deposition problem. If you avoid sulfates but keep using silicones, the buildup becomes progressively worse. This is why so many curly-haired women feel trapped in a cycle of products that don’t work. The chemistry is working against them.
Cationic Polymers and Quaternary Compounds: The Buildup Magnets
Polyquaterniums (Polyquaternium-7, -10, -11), Guar Hydroxypropyltrimonium Chloride, Behentrimonium Chloride. These ingredients are conditioning agents that carry a positive charge. They’re attracted to the negative charge on damaged hair. Sounds helpful. In hard water, it creates a buildup nightmare.
The positive charge that makes these ingredients effective also makes them bind strongly to mineral deposits. Calcium and magnesium ions are also positively charged. When cationic polymers encounter hard water minerals on your hair, they form complexes that are extremely difficult to remove. The result is a sticky, heavy coating that weighs down curls and blocks moisture.
Research from the Personal Care Products Council indicates that quaternary ammonium compounds show significantly increased deposition in the presence of hard water minerals. This isn’t a minor effect. The deposition rate can increase by 300% or more compared to soft water conditions.
You’ll notice this as a progressive loss of curl definition. Your hair starts to feel coated and sticky, especially near the roots where product application is heaviest. Curls that used to spring back when stretched now stay elongated. The weight of the buildup is literally pulling your curl pattern down. A chelating shampoo like Regrowth+ can help remove this accumulated mineral-polymer complex before it causes permanent damage to your curl structure.
 Learning to spot these ingredients quickly becomes second nature. Look for them in the first five ingredients listed, where concentration is highest.
Learning to spot these ingredients quickly becomes second nature. Look for them in the first five ingredients listed, where concentration is highest.
Waxes and Heavy Oils: When Moisture Becomes a Barrier
Petrolatum, Mineral Oil, Beeswax, Lanolin. These occlusive ingredients create a moisture barrier on your hair. In some climates, this can be protective. In hard water regions, they seal in the wrong things.
The problem is timing and chemistry. When you apply a heavy wax or oil-based product to hair that’s been washed in hard water, you’re sealing mineral deposits against your hair shaft. These occlusives don’t discriminate. They trap whatever is already on your hair, including calcium and magnesium buildup.
This is particularly problematic with leave-in conditioners and curl creams that contain these ingredients. You apply them to damp hair fresh from a hard water rinse. The minerals are still wet and actively bonding to your hair shaft. Then you seal them in with an occlusive layer. By the time your hair dries, the damage is locked in place.
Heavy oils like castor oil and shea butter can have a similar effect, though they’re not as completely occlusive as petrolatum. In high humidity environments common in the Gulf region, these heavy ingredients also attract atmospheric moisture, which in turn attracts more mineral deposits from your next wash. It becomes a self-perpetuating cycle of buildup.
Proteins in Hard Water: When Strengthening Becomes Stiffening
Hydrolyzed wheat protein, keratin, silk amino acids. Protein treatments are essential for maintaining curl structure and preventing breakage. But in hard water conditions, they require careful management.
Proteins work by temporarily filling in gaps in your hair’s structure, reinforcing weak points in the cuticle and cortex. When mineral buildup is present, proteins can bind to those minerals instead of to your hair. The result is a stiff, brittle coating rather than strengthened hair. Your curls feel like wire instead of elastic.
This doesn’t mean you should avoid proteins entirely. It means you need to chelate before protein treatments. If you apply a protein mask to hair that’s coated in mineral deposits, you’re essentially gluing the minerals to your hair shaft. The protein bonds with the calcium and magnesium, creating a rigid structure that makes your hair more prone to breakage, not less.
Signs of protein-mineral overload: hair that snaps easily when stretched, curls that have lost all elasticity, a crunchy texture that doesn’t soften even after conditioning. If you’re experiencing these symptoms and you’ve been using protein treatments regularly, the issue isn’t too much protein. It’s protein interacting with hard water minerals. You need to remove the mineral layer first, then reassess your protein needs.
What to Look for Instead: Hard Water-Safe Alternatives
Gentle sulfate-free cleansers: Look for Cocamidopropyl Betaine, Decyl Glucoside, or Sodium Cocoyl Isethionate. These surfactants clean effectively without stripping your hair’s protective oils, and they don’t interact as strongly with hard water minerals. They’re not completely immune to mineral interference, but the buildup is significantly less severe.
Water-soluble silicones: If you want the smoothing benefits of silicones without the buildup, choose ingredients that end in ‘-ol’ rather than ‘-one.’ Dimethiconol, for example, is water-soluble and rinses away cleanly even in hard water. PEG-modified silicones (anything with ‘PEG’ in the name followed by a silicone) are also water-soluble and safer for regular use.
Light, penetrating oils: Argan oil, jojoba oil, and grapeseed oil have smaller molecular structures that can penetrate the hair shaft rather than just coating it. They’re less likely to seal in mineral deposits because they’re absorbed before the minerals fully bond to your hair. Use them on soaking wet hair immediately after washing for best results.
Chelating agents: EDTA (Ethylenediaminetetraacetic acid), phytic acid, and citric acid actively bind to hard water minerals and help remove them from your hair. Look for these ingredients in your shampoo or use a dedicated chelating treatment weekly. This is the most important category for anyone dealing with hard water. Without regular chelation, every other product choice becomes less effective.
Reading Labels: Where Problem Ingredients Hide
Ingredient lists are ordered by concentration. The first five ingredients make up the majority of the product. This is where you need to focus your attention. If a problematic ingredient appears in the first five, it’s present in high enough concentration to cause issues in hard water conditions.
Watch for ingredient synonyms. Sulfates hide under names like Sodium Myreth Sulfate, Ammonium Lauryl Sulfate, and TEA-Lauryl Sulfate. They’re all sulfates. Silicones often end in ‘-cone,’ ‘-conol,’ or ‘-siloxane.’ If you see these patterns in the first half of the ingredient list, the product isn’t suitable for hard water use.
Fragrance and parfum are catch-all terms that can hide problematic ingredients. While not all fragrances cause buildup, they’re not required to disclose their components. If you’re experiencing unexplained buildup with products that appear clean otherwise, fragrance components may be interacting with your water minerals.
A useful resource for ingredient checking is the Environmental Working Group’s Skin Deep database. While it focuses primarily on safety rather than hard water compatibility, it provides detailed breakdowns of what each ingredient does and what it’s chemically similar to. This can help you identify problem ingredients even when they appear under unfamiliar names.
References
- Effects of Hard Water on Hair and Scalp: Interaction with Surfactants - Journal of Cosmetic Science
- Calcium and Magnesium Precipitation with Anionic Surfactants - Colloids and Surfaces B: Biointerfaces
- Cationic Polymers in Hair Care: Deposition and Performance - Personal Care Products Council
- Cosmetic Ingredient Safety Database - Environmental Working Group
